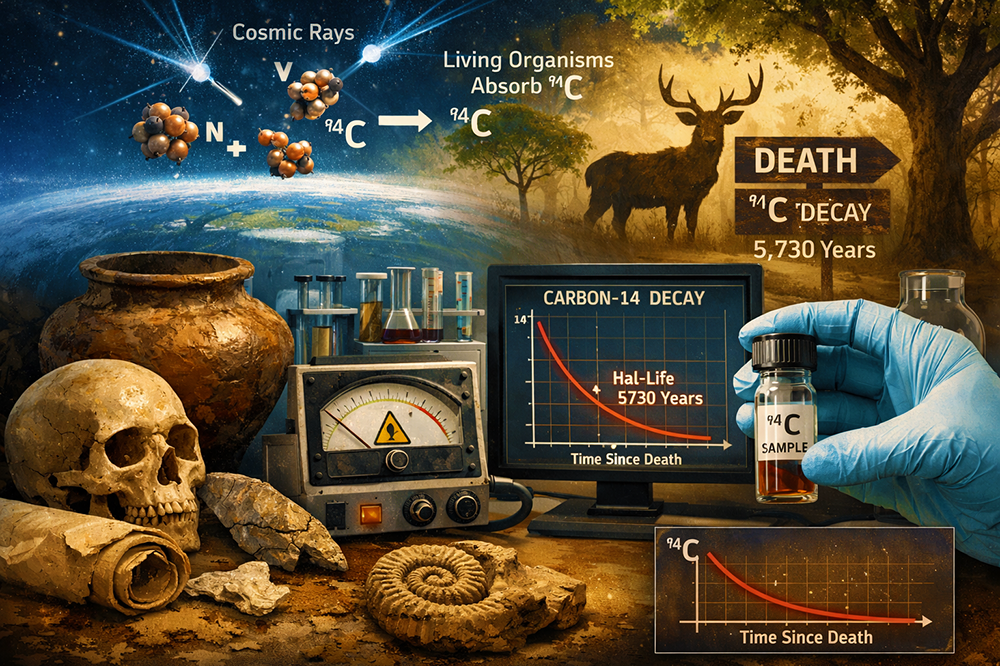
Carbon dating, also known as radiocarbon dating, is a scientific method used to determine the age of once-living materials such as wood, bone, cloth, and charcoal. It is based on the radioactive isotope carbon-14 (¹⁴C), which forms in the upper atmosphere when cosmic rays interact with nitrogen. Living organisms continuously absorb carbon, including a small amount of carbon-14, through processes like breathing and eating. However, when an organism dies, it stops taking in carbon, and the carbon-14 it contains begins to decay at a known rate, with a half-life of about 5,730 years. By measuring the remaining amount of carbon-14 in a sample and comparing it to the expected original level, scientists can estimate how long it has been since the organism died. Carbon dating has revolutionized archaeology, paleontology, and environmental science, helping researchers date ancient artifacts, fossils, and historical objects up to around 50,000 years old with remarkable accuracy.
Whoitam Software and Technology ⟬ 2014-2026 ⟭. Site Design by MD.FEROSE ⇵